The lower the l quantum number of an orbital, the more time the electron spends penetrating near the nucleus. Shielding results in lessening this effective nuclear charge on outer shell electrons, since they spend most of their time at distances farther from the nucleus and are shielded from the nuclear charge by the inner electrons. 8.9 Penetration occurs when the probability distribution of an orbital is large near the nucleus, which results in an increase of the overall attraction of the nucleus for the electron, lowering its energy. As the number of inner electrons increases, shielding increases, and the effective nuclear charge decreases. The effective nuclear charge is the nuclear charge an electron actually experiences. 8.8 Shielding occurs when inner electrons protect or shield outer electrons from the full nuclear attractive force. Be 3+ would be more like H since both have only one 1s electron. This splitting is due to electron-electron repulsions. In many electron systems, the principal energy levels are split into sublevels of differing energies. 8.7 In a one-electron system, all sublevels of a particular level (such as 2s and 2p) have the same energy. Within a particular orbital, there can be only two electrons and they must have opposing spins. 8.6 The exclusion principle states that no two electrons in the same atom may have the same four quantum numbers. (l – 1), +l s : –1/2 or +1/2 8.5 The quantum number m s relates to just the electron all the others describe the orbital. n – 1 The allowed values of m l The allowed values of m : integers from –l to 0 to +l: –l, (–l + 1). a) Predicted atomic mass (K) = Solution: Na Rb 2 + = 22.99 85.47 2 + = 54.23 amu (actual value = 39.10 amu) b) Predicted melting point (Br 2) = 2 2 Cl I 2 + = 101.0 113.6 2 − + = 6.3☌ (actual value = –7.2☌) 8.4 The allowed values of n: positive integers: 1, 2, 3, 4.∞ The allowed values of l: integers from 0 to n – 1: 0, 1, 2. 8.3 Plan: The value should be the average of the elements above and below the one of interest. One of these exceptions is iodine, Z = 53, which is after tellurium, Z = 52, even though tellurium has a higher atomic mass. This makes a difference in the sequence of elements in only a few cases, as the larger atomic number usually has the larger atomic mass. 8.2 Today, the elements are listed in order of increasing atomic number. 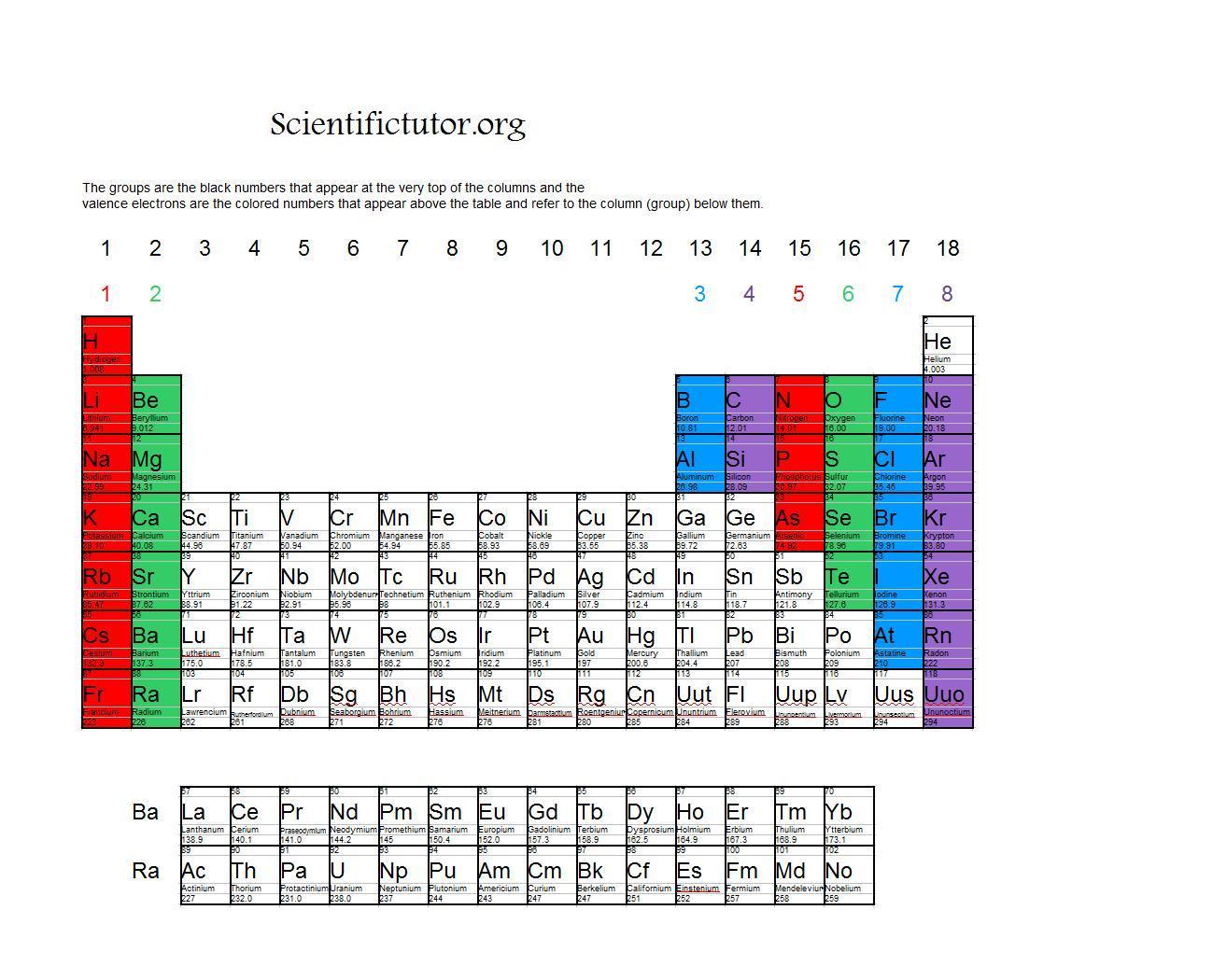
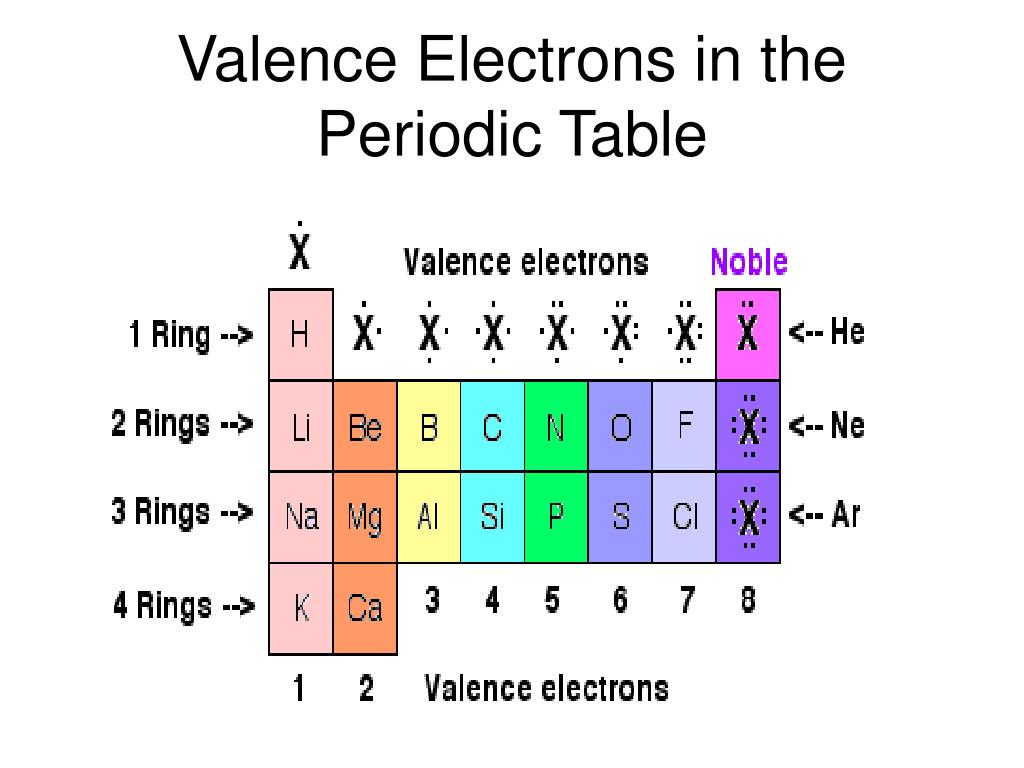
The theoretical basis for the table in terms of atomic number and electron configuration does not allow for an " unknown element " between Sn and Sb. So, sodium should contain 11 electrons in its electron configuration and magnesium should contain 12.ĮND–OF–CHAPTER PROBLEMS 8.1 Elements are listed in the periodic table in an ordered, systematic way that correlates with a periodicity of their chemical and physical properties. For example, sodium has an atomic number of 11 and magnesium has an atomic number of 12. The atomic number of an atom in the ground state is the same as the number of electrons. Each atom has its own position on the periodic table. Electron Configuration An electron configuration is the arrangement of electrons around the nucleus of an atom. Examples: Magnesium's ground state electron configuration is 1s 2 2s 2 p 6 3s 2, the valence electrons would be the 3s electrons because 3 is the highest principle quantum number. When an electron loses energy, then it can move to an inner shell which is not fully occupied. Or the electron can even break free from its associated atom's valence shell this is ionization to form a positive ion. An energy gain can trigger an electron to move (jump) to an outer shell this is known as atomic excitation. Similar to an electron in an inner shell, a valence electron has the ability to absorb or release energy in the form of a photon. The simplest way to identify the valence electrons is to look for the highest number in the electron configuration of an atom (the principle quantum number). They are typically the electrons with the highest value of the principle quantum number, n. A valence electron is an electron that is the most likely to be involved in a chemical reaction. For a main group element, a valence electron can exist only in the outermost electron shell in a transition metal, a valence electron can also be in an inner shell. The presence of valence electrons can determine the element's chemical properties, such as its valence-whether it may bond with other elements and, if so, how readily and with how many. Valence Electron Definition Valence electron is an electron in one of the outer shells of an atom that can participate in forming chemical bonds with other atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
